What Leads to the Formation of an Ionic Bond With Hg22+?

Ionic bonding is a type of chemical bonding that involves the electrostatic allure between oppositely charged ions, or betwixt two atoms with sharply different electronegativities,[1] and is the master interaction occurring in ionic compounds. It is one of the main types of bonding along with covalent bonding and metallic bonding. Ions are atoms (or groups of atoms) with an electrostatic charge. Atoms that gain electrons brand negatively charged ions (called anions). Atoms that lose electrons brand positively charged ions (called cations). This transfer of electrons is known as electrovalence in dissimilarity to covalence. In the simplest case, the cation is a metallic cantlet and the anion is a nonmetal atom, but these ions can be of a more complex nature, e.m. molecular ions like NH +
4 or SO ii−
4 . In simpler words, an ionic bond results from the transfer of electrons from a metallic to a non-metal in order to obtain a full valence shell for both atoms.
Information technology is important to recognize that clean ionic bonding — in which 1 atom or molecule completely transfers an electron to some other — cannot exist: all ionic compounds have some degree of covalent bonding, or electron sharing. Thus, the term "ionic bonding" is given when the ionic graphic symbol is greater than the covalent character – that is, a bond in which a big electronegativity difference exists between the two atoms, causing the bonding to be more polar (ionic) than in covalent bonding where electrons are shared more equally. Bonds with partially ionic and partially covalent character are called polar covalent bonds.
Ionic compounds conduct electricity when molten or in solution, typically not when solid. Ionic compounds generally have a high melting bespeak, depending on the charge of the ions they consist of. The higher the charges the stronger the cohesive forces and the higher the melting bespeak. They likewise tend to be soluble in water; the stronger the cohesive forces, the lower the solubility.[2]
Overview [edit]
Atoms that have an almost full or almost empty valence beat tend to be very reactive. Atoms that are strongly electronegative (as is the case with halogens) often take only one or ii empty orbitals in their valence shell, and frequently bail with other molecules or gain electrons to grade anions. Atoms that are weakly electronegative (such equally alkali metals) have relatively few valence electrons, which can easily exist shared with atoms that are strongly electronegative. As a issue, weakly electronegative atoms tend to distort their electron cloud and form cations.
Formation [edit]
Ionic bonding can result from a redox reaction when atoms of an element (usually metallic), whose ionization energy is depression, give some of their electrons to achieve a stable electron configuration. In doing and then, cations are formed. An cantlet of another element (usually nonmetal) with greater electron affinity accepts 1 or more than electrons to attain a stable electron configuration, and afterwards accepting electrons an atom becomes an anion. Typically, the stable electron configuration is one of the noble gases for elements in the s-cake and the p-cake, and particular stable electron configurations for d-block and f-block elements. The electrostatic attraction between the anions and cations leads to the formation of a solid with a crystallographic lattice in which the ions are stacked in an alternating way. In such a lattice, it is unremarkably non possible to distinguish discrete molecular units, so that the compounds formed are not molecular in nature. Still, the ions themselves can exist complex and form molecular ions like the acetate anion or the ammonium cation.

Representation of ionic bonding betwixt lithium and fluorine to form lithium fluoride. Lithium has a depression ionization free energy and readily gives up its lone valence electron to a fluorine atom, which has a positive electron affinity and accepts the electron that was donated by the lithium atom. The end-result is that lithium is isoelectronic with helium and fluorine is isoelectronic with neon. Electrostatic interaction occurs between the two resulting ions, just typically assemblage is not limited to two of them. Instead, aggregation into a whole lattice held together by ionic bonding is the result.
For example, common common salt is sodium chloride. When sodium (Na) and chlorine (Cl) are combined, the sodium atoms each lose an electron, forming cations (Na+), and the chlorine atoms each gain an electron to form anions (Cl−). These ions are and so attracted to each other in a 1:1 ratio to form sodium chloride (NaCl).
- Na + Cl → Na+ + Cl− → NaCl
However, to maintain charge neutrality, strict ratios betwixt anions and cations are observed so that ionic compounds, in general, obey the rules of stoichiometry despite not beingness molecular compounds. For compounds that are transitional to the alloys and possess mixed ionic and metallic bonding, this may non be the case anymore. Many sulfides, e.g., do grade non-stoichiometric compounds.
Many ionic compounds are referred to equally salts as they tin can also be formed by the neutralization reaction of an Arrhenius base like NaOH with an Arrhenius acrid similar HCl
- NaOH + HCl → NaCl + HiiO
The table salt NaCl is so said to consist of the acid residuum Cl− and the base rest Na+.
The removal of electrons to class the cation is endothermic, raising the system's overall free energy. There may also be energy changes associated with breaking of existing bonds or the addition of more than 1 electron to class anions. However, the activity of the anion's accepting the cation'southward valence electrons and the subsequent attraction of the ions to each other releases (lattice) energy and, thus, lowers the overall energy of the arrangement.
Ionic bonding volition occur only if the overall energy modify for the reaction is favorable. In general, the reaction is exothermic, merely, e.m., the formation of mercuric oxide (HgO) is endothermic. The accuse of the resulting ions is a major factor in the strength of ionic bonding, e.g. a salt C+A− is held together past electrostatic forces roughly four times weaker than C2+A2− according to Coulomb's law, where C and A represent a generic cation and anion respectively. The sizes of the ions and the item packing of the lattice are ignored in this rather simplistic statement.
Structures [edit]
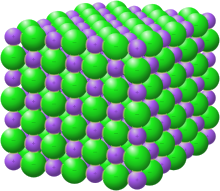
In the stone salt lattice, each sodium ion (purple sphere) has an electrostatic interaction with its eight nearest-neighbour chloride ions (green spheres)
Ionic compounds in the solid state form lattice structures. The ii chief factors in determining the form of the lattice are the relative charges of the ions and their relative sizes. Some structures are adopted by a number of compounds; for example, the structure of the stone salt sodium chloride is besides adopted by many brine halides, and binary oxides such as magnesium oxide. Pauling'due south rules provide guidelines for predicting and rationalizing the crystal structures of ionic crystals
Strength of the bonding [edit]
For a solid crystalline ionic chemical compound the enthalpy change in forming the solid from gaseous ions is termed the lattice energy. The experimental value for the lattice free energy can be adamant using the Born–Haber cycle. It can as well be calculated (predicted) using the Born–Landé equation as the sum of the electrostatic potential energy, calculated by summing interactions between cations and anions, and a brusque-range repulsive potential free energy term. The electrostatic potential can be expressed in terms of the interionic separation and a constant (Madelung constant) that takes business relationship of the geometry of the crystal. The further away from the nucleus the weaker the shield. The Built-in-Landé equation gives a reasonable fit to the lattice free energy of, e.chiliad., sodium chloride, where the calculated (predicted) value is −756 kJ/mol, which compares to −787 kJ/mol using the Born–Haber cycle.[three] [4] In aqueous solution the binding strength can be described by the Bjerrum or Fuoss equation every bit role of the ion charges, rather independent of the nature of the ions such as polarizability or size [v] The strength of salt bridges is most often evaluated by measurements of equilibria between molecules containing cationic and anionic sites, most often in solution. [6] Equilibrium constants in water signal additive gratuitous free energy contributions for each salt bridge. Another method for the identification of hydrogen bonds also in complicated molecules is crystallography, sometimes also NMR-spectroscopy.
The bonny forces defining the strength of ionic bonding can be modeled by Coulomb's Law. Ionic bail strengths are typically (cited ranges vary) between 170 and 1500 kJ/mol.[7] [8]
Polarization ability effects [edit]
Ions in crystal lattices of purely ionic compounds are spherical; nevertheless, if the positive ion is small and/or highly charged, it will distort the electron cloud of the negative ion, an effect summarised in Fajans' rules. This polarization of the negative ion leads to a build-up of extra accuse density between the two nuclei, that is, to partial covalency. Larger negative ions are more easily polarized, but the effect is commonly important only when positive ions with charges of 3+ (e.g., Al3+) are involved. However, 2+ ions (Beii+) or even i+ (Li+) show some polarizing power because their sizes are so small (due east.g., LiI is ionic but has some covalent bonding present). Note that this is not the ionic polarization event that refers to displacement of ions in the lattice due to the application of an electric field.
Comparing with covalent bonding [edit]
In ionic bonding, the atoms are spring by allure of oppositely charged ions, whereas, in covalent bonding, atoms are bound by sharing electrons to attain stable electron configurations. In covalent bonding, the molecular geometry around each cantlet is determined past valence trounce electron pair repulsion VSEPR rules, whereas, in ionic materials, the geometry follows maximum packing rules. One could say that covalent bonding is more directional in the sense that the energy penalization for not adhering to the optimum bail angles is large, whereas ionic bonding has no such punishment. In that location are no shared electron pairs to repel each other, the ions should just be packed every bit efficiently every bit possible. This ofttimes leads to much higher coordination numbers. In NaCl, each ion has half-dozen bonds and all bond angles are 90°. In CsCl the coordination number is 8. By comparison carbon typically has a maximum of four bonds.
Purely ionic bonding cannot exist, every bit the proximity of the entities involved in the bonding allows some degree of sharing electron density between them. Therefore, all ionic bonding has some covalent character. Thus, bonding is considered ionic where the ionic grapheme is greater than the covalent grapheme. The larger the deviation in electronegativity between the ii types of atoms involved in the bonding, the more ionic (polar) it is. Bonds with partially ionic and partially covalent character are called polar covalent bonds. For example, Na–Cl and Mg–O interactions have a few percentage covalency, while Si–O bonds are usually ~50% ionic and ~50% covalent. Pauling estimated that an electronegativity difference of one.7 (on the Pauling scale) corresponds to 50% ionic character, so that a deviation greater than 1.seven corresponds to a bond which is predominantly ionic.[nine]
Ionic grapheme in covalent bonds tin be direct measured for atoms having quadrupolar nuclei (2H, fourteenN, 81,79Br, 35,37Cl or 127I). These nuclei are generally objects of NQR nuclear quadrupole resonance and NMR nuclear magnetic resonance studies. Interactions between the nuclear quadrupole moments Q and the electric field gradients (EFG) are characterized via the nuclear quadrupole coupling constants
- QCC = e two q zz Q / h
where the eq zz term corresponds to the principal component of the EFG tensor and e is the unproblematic charge. In turn, the electric field gradient opens the mode to clarification of bonding modes in molecules when the QCC values are accurately determined by NMR or NQR methods.
In general, when ionic bonding occurs in the solid (or liquid) state, it is not possible to talk about a single "ionic bond" between 2 individual atoms, because the cohesive forces that keep the lattice together are of a more collective nature. This is quite different in the case of covalent bonding, where we can oftentimes speak of a distinct bail localized betwixt two item atoms. Yet, even if ionic bonding is combined with some covalency, the upshot is non necessarily discrete bonds of a localized character. In such cases, the resulting bonding ofttimes requires description in terms of a band construction consisting of gigantic molecular orbitals spanning the unabridged crystal. Thus, the bonding in the solid often retains its collective rather than localized nature. When the difference in electronegativity is decreased, the bonding may then pb to a semiconductor, a semimetal or eventually a metallic conductor with metallic bonding.
Encounter also [edit]
- Coulomb's law
- Salt span (protein and supramolecular)
- Ionic potential
- Linear combination of atomic orbitals
- Hybridization
- Chemical polarity
- Ioliomics
- Electron configuration
- Aufbau principle
- Quantum numbers
- Azimuthal breakthrough number
- Principal quantum number
- Magnetic breakthrough number
- Spin quantum number
References [edit]
- ^ "Ionic bond". IUPAC Compendium of Chemical Terminology. 2009. doi:ten.1351/goldbook.IT07058. ISBN978-0-9678550-9-7.
- ^ Schneider, Hans-Jörg (2012). "Ionic Interactions in Supramolecular Complexes". Ionic Interactions in Natural and Synthetic Macromolecules. pp. 35–47. doi:10.1002/9781118165850.ch2. ISBN9781118165850.
- ^ David Arthur Johnson, Metals and Chemical Change, Open University, Purple Society of Chemistry, 2002, ISBN 0-85404-665-8
- ^ Linus Pauling, The Nature of the Chemical Bond and the Structure of Molecules and Crystals: An Introduction to Modernistic Structural Chemical science, Cornell Academy Press, 1960 ISBN 0-801-40333-ii doi:10.1021/ja01355a027
- ^ Schneider, H.-J.; Yatsimirsky, A. (2000) Principles and Methods in Supramolecular Chemistry. Wiley ISBN 9780471972532
- ^ Biedermann F, Schneider HJ (May 2016). "Experimental Binding Energies in Supramolecular Complexes". Chemical Reviews. 116 (9): 5216–300. doi:10.1021/acs.chemrev.5b00583. PMID 27136957.
- ^ Soboyejo, West.O (2003). Mechanical properties of engineered materials. Marcel Dekker. pp. 16–17. ISBN 0-203-91039-7. OCLC 54091550.
- ^ Askeland, Donald R. (January 2015). The science and engineering of materials. Wright, Wendelin J. (Seventh ed.). Boston, MA. pp. 38. ISBN 978-1-305-07676-1. OCLC 903959750.
- ^ L. Pauling The Nature of the Chemical Bail (tertiary ed., Oxford Academy Press 1960) p.98-100.
External links [edit]
- Ionic bonding tutorial
- Video on ionic bonding
Source: https://en.wikipedia.org/wiki/Ionic_bonding
0 Response to "What Leads to the Formation of an Ionic Bond With Hg22+?"
Post a Comment